AI Continues to Build the Foundation for a Remarkable Future in Biology


July 28, 2022, was a historic day in both biology and artificial intelligence (AI). DeepMind, an Alphabet-owned AI research firm, made the structural data on more than 200 million proteins from its AlphaFold tool freely available. This represents data on roughly 1 million species and covers the vast majority of known proteins on Earth.1
In Proteins, Shape Can Determine Function
In the late 1990s into the early 2000s, the scientific community was awash with news of the race to sequence the human genome. This genome contains the instructions embedded in DNA about how cells should build certain structures, typically by forming proteins made from different combinations of amino acids.
In a sense, DNA is the instruction manual, amino acids are the building blocks and proteins are the product. Knowing the code, though important, is not enough.
Figure 1 is instructive on the point. This is the image of a protein that may protect the organism responsible for malaria from attack by the human immune system. Even if you knew the list and the order of all the amino acids, it would be difficult to go from that list to something that looks like Figure 1 in three dimensions.
Figure 1: Protein Associated with the Malaria Parasite
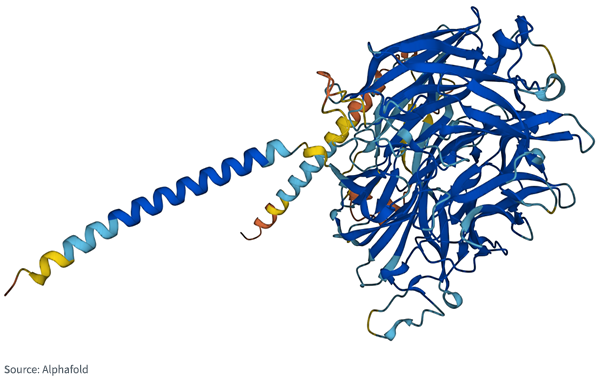
The importance of the shape of the protein can not be overstated:
- It can correspond to the way in which it might react in the presence of different molecules, like those associated with various drug therapies.
- Variations in the shape—sometimes termed mutations—could be instructive in determining the causal factors of certain symptoms or diseases.
- Parts of the shape could be used as targets. Think of the “spike protein” associated with the virus behind the COVID-19 pandemic, specifically targeted in the mRNA vaccines.
AlphaFold Represents a Leap Forward
Scientific breakthroughs are difficult, in that, in many cases, one builds on another and another and another. The process can take decades before widespread results impact the lives of the general public. For instance, we sequenced the human genome, but that did not necessarily lead to immediate cures for all sorts of diseases or conditions. While mRNA research had been occurring for decades, the COVID-19 pandemic was a catalyst that supercharged the process of using mRNA in the specific case of the vaccines.
AlphaFold’s new database is therefore unlikely to lead to immediate cures for challenging conditions. The critical element regards how researchers who would have formerly had to undertake a cumbersome process of X-ray crystallography to determine the shape of a given protein could instead go to the database. Experimental techniques would still have their place, but less time would have to be spent on the equivalent of the blank page.
What’s also incredible is that AlphaFold’s database is, in conjunction with the European Molecular Biology Laboratory’s European Bioinformatics Institute (EMBL-EBI), freely available with a simple interface. It also provides an estimate of the accuracy, recognizing that predictions based on AI do not always yield perfect results. Roughly 35% of the 214 million predictions are deemed highly accurate—roughly as good as experimental results. A further 45% are deemed to be accurate enough for many applications.2
Drug Discovery: Better Therapeutics Developed More Efficiently
Even before the onset of inflation at the levels seen in the summer of 2022, it was widely recognized that drug development is time-consuming and expensive. As a result, many medications carry exorbitant price tags. Any process that could mitigate this pressure without degrading the quality of the therapies would be enormously valuable.
Considering the following could be instructive as the space continues to progress:3
- Pipeline Growth: From 2010 to 2021, 20 smaller companies focused on AI drug discovery, typically with a focus on smaller molecules, had development pipelines that were roughly 50% as robust as those of 20 of the largest big pharma companies. We recognize that the reporting of pipelines may not be perfect and that a molecule in a pipeline is not a finished product, but activity is the first step on the path.
- Pipeline Composition: This is not always disclosed, but the information available indicates that AI-focused companies tend to concentrate on well-established biological targets for their therapies, around which much is known. Data is the fuel for AI, and these companies will also want higher chances of success. Bigger pharma companies will be more likely to venture into more emerging areas of drug discovery.
- Chemical Structures and Properties: It is too early to draw any robust conclusions regarding AI drug discovery efforts versus big pharma efforts on this point.
- Discovery Timelines: Preliminary data appears to indicate that, while traditional approaches tend to take five or six years in preclinical phases, AI-focused drug discovery might be able to, in certain cases, take this timeline down to four years.
We’d note that it’s more a story of progress than perfection, in that we appear to be some distance away from AI being able to fully create new drugs. But AI represents an entirely new set of tools that could have beneficial impacts. AlphaFold’s database, for example, may provide drug researchers with important inputs and catalysts for different ideas, even if it doesn’t have the immediate answer or cure right there in its system.
Focus on the AI and BioRevolution Megatrends
At WisdomTree, we focus on both the AI megatrend and the BioRevolution megatrend. What we see here with the case of AlphaFold is an important case study in the fact that AI is a tool that can potentially supercharge other megatrends, in this case the BioRevolution. It is no accident that the BioRevolution is ramping up at the same time massive amounts of data, massive amounts of processing power and other things like cloud computing are readily available. It is exciting to consider what the coming decades can bring in these areas.
1 Source: Callaway, Ewen, “’The Entire Protein Universe’: AI Predicts Shape of Nearly Every Known Protein,” Nature, Volume 608, 8/4/22.
2 Source: Callaway, 8/4/22.
3 Source: Jayatunga et al, “AI in small-molecule drug discovery: a coming wave?” Nature Review: Drug Discovery, Volume 21, 3/22.
Important Risks Related to this Article
Christopher Gannatti is an employee of WisdomTree UK Limited, a European subsidiary of WisdomTree Asset Management, Inc.’s parent company, WisdomTree Investments, Inc.
DeepMind is a subsidiary of Alphabet. As of August 16, 2022, Alphabet was a 1.36% exposure in WTAI and a 0% exposure in WDNA.
WTAI: There are risks associated with investing, including possible loss of principal. The Fund invests in companies primarily involved in the investment theme of Artificial Intelligence (AI) and Innovation. Companies engaged in AI typically face intense competition and potentially rapid product obsolescence. These companies are also heavily dependent on intellectual property rights and may be adversely affected by loss or impairment of those rights. Additionally, AI companies typically invest significant amounts of spending on research and development, and there is no guarantee that the products or services produced by these companies will be successful. Companies that are capitalizing on Innovation and developing technologies to displace older technologies or create new markets may not be successful. The Fund invests in the securities included in, or representative of, its Index regardless of their investment merit and the Fund does not attempt to outperform its Index or take defensive positions in declining markets. The composition of the Index is governed by an Index Committee and the Index may not perform as intended. Please read the Fund’s prospectus for specific details regarding the Fund’s risk profile.
WDNA: There are risks associated with investing, including possible loss of principal. The Fund invests in BioRevolution companies, which are companies significantly transformed by advancements in genetics and biotechnology. BioRevolution companies face intense competition and potentially rapid product obsolescence. These companies may be adversely affected by the loss or impairment of intellectual property rights and other proprietary information or changes in government regulations or policies. Additionally, BioRevolution companies may be subject to risks associated with genetic analysis. The Fund invests in the securities included in, or representative of, its Index regardless of their investment merit and the Fund does not attempt to outperform its Index or take defensive positions in declining markets. The composition of the Index is governed by an Index Committee and the Index may not perform as intended. Please read the Fund’s prospectus for specific details regarding the Fund’s risk profile.

Christopher Gannatti began at WisdomTree as a Research Analyst in December 2010, working directly with Jeremy Schwartz, CFA®, Director of Research. In January of 2014, he was promoted to Associate Director of Research where he was responsible to lead different groups of analysts and strategists within the broader Research team at WisdomTree. In February of 2018, Christopher was promoted to Head of Research, Europe, where he was based out of WisdomTree’s London office and was responsible for the full WisdomTree research effort within the European market, as well as supporting the UCITs platform globally. In November 2021, Christopher was promoted to Global Head of Research, now responsible for numerous communications on investment strategy globally, particularly in the thematic equity space. Christopher came to WisdomTree from Lord Abbett, where he worked for four and a half years as a Regional Consultant. He received his MBA in Quantitative Finance, Accounting, and Economics from NYU’s Stern School of Business in 2010, and he received his bachelor’s degree from Colgate University in Economics in 2006. Christopher is a holder of the Chartered Financial Analyst Designation.

